- ⌂
-
Le DPM
Les Equipements
L'Environnement
Zoom sur... Le Bâtiment André Rassat
Nommé d'après une figure tutélaire de la chimie grenobloise, ce bâtiment est recouvert d’une double peau en feuille métallique qui apporte une protection thermique sur 3 côtés et crée une unité architecturale favorisant l'intégration parmi les arbres du site.
-
Thématiques
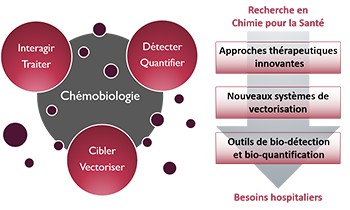
Le Thème
« Approches thérapeutiques innovantes »De nouvelles solutions thérapeutiques, de la cible biomacromolécu-laire émergente aux approches originales pour traiter les maladies
Le Thème
« Nouveaux systèmes de vectorisation »Combiner les propriétés d’inclusion de principes actifs, de franchisse-ment de barrières, d’adressage et de relar-gage en milieu vivant
Le Thème
« Outils de bio-détection et bio-quantification »Des dispositifs analytiques originaux pour la détection de cibles, de l’ion au micro-organisme en milieu complexe
Zoom sur... La Chémobiologie
-
Équipes
L'Équipe « COMET »
« COMET » développe la conception rationnelle, la synthèse et/ou l'extraction de composés à forte diversité/complexité comme nouveaux agents thérapeutiques et outils moléculaires pour la pénétration cellulaire ou la détection de biomolécules, actifs in vivo.L'Équipe « NOVA »
« NOVA » utilise des acides nucléiques fonctionnels comme éléments de reconnaissance pour des applications thérapeutiques ou diagnostiques, comme la sélection d'oligonucléotides, ou le développement de dispositifs d'analyses et de nanovecteurs.Les Services
-
Productions
Les Publications
La Vulgarisation
Les JSM
Zoom sur... La 12ème JSM (15 juin 2023)
Le DPM organise des journées scientifiques consacrées au médicament. L'objectif est de rassembler les spécialistes académiques et industriels autour d'une thématique. 2023 : Apports de la Chimie Click et de la Lumière en Chemobiologie
-
Partenariats
Les Formations
Les Consortiums
Les Financements
Zoom sur... L'environnement Grenoblois
Le DPM est un acteur central sur le bassin grenoblois en chimie, biologie et santé, lié au CHU Grenoble Alpes et à de nombreuses autres organisations : Pole de Recherche CBS, ICMG, Labex ARCANE, EUR CBH, Institut Carnot Polynat, Réseau GREEN.
Article
- Projet
- Ahcène BOUMENDJEL, Marine PEUCHMAUR, Romain HAUDECOEUR, Hoang Truc Phuong NGUYEN,
- Titre
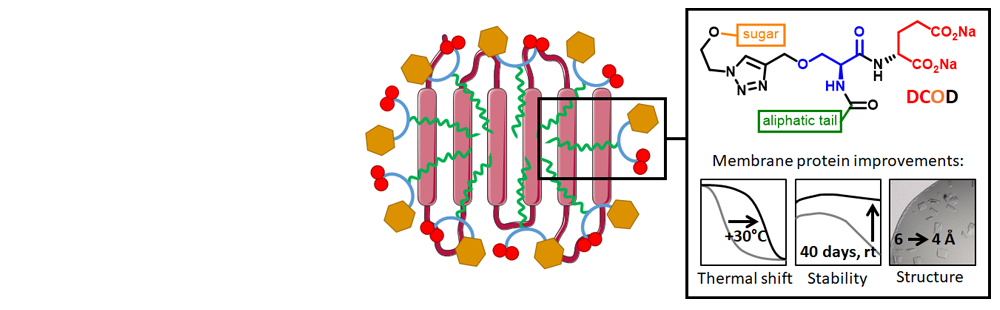
- Glycosyl-Substituted Dicarboxylates as Detergents for the Extraction, Overstabilization, and Crystallization of Membrane Proteins
-
[Full paper
 ]
] - Auteurs
- K.-A. Nguyen, M. Peuchmaur, S. Magnard, R. Haudecoeur, C. Boyère, S. Mounien, I. Benammar, V. Zampieri, S. Igonet, V. Chaptal, A. Jawhari, A. Boumendjel, P. Falson
- Edition
- Angew. Chem. Int. Ed. Engl. 2018, 57, 2948-2952.
- Année
- 2018
- Résumé
- To tackle the problem of membrane proteins (MPs) instability in detergent solutions, we designed a series of dicarboxylate-oside detergents (DCODs) in which we optimized the polar head to clamp the membrane domain, including on one side a cluster of two carboxyl groups that promotes salt-bridges with basic residues abundant at the membrane-cytoplasm interface of MPs, and on the other side a sugar to generate H-bonds. Tested on BmrA, an ATP-binding cassette pump, DCODs 8b, 8c, and 9b preserved its ATPase function upon extraction much more efficiently than reference or recently designed detergents. DCOD 8b also conferred a long-range functional stability of DDM-purified BmrA of up to 40 days at 18 °C. DCODs 8(a,b,f) and 9(a,b) induced a thermal shift of 20 to 29°C for BmrA and of 13 to 21°C for the native version of the G-protein coupled adenosine receptor A2AR. The same proteins tested by size-exclusion chromatography with DCOD 8b behaved as with DDM. Finally, DCOD 8f and 8g improved the diffraction of BmrA crystals from 6 to 4 Å. These results make DCODs promising and powerful tools for the structural biology of MPs.




 Annuaire
Annuaire Contact
Contact Plan d'accès
Plan d'accès ENG
ENG Login
Login


