- ⌂
-
Le DPM
Les Equipements
L'Environnement
Zoom sur... Le Bâtiment André Rassat
Nommé d'après une figure tutélaire de la chimie grenobloise, ce bâtiment est recouvert d’une double peau en feuille métallique qui apporte une protection thermique sur 3 côtés et crée une unité architecturale favorisant l'intégration parmi les arbres du site.
-
Thématiques
Le Thème
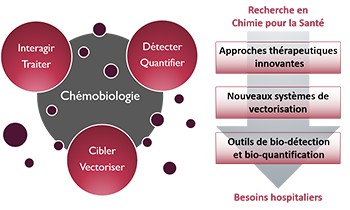
« Approches thérapeutiques innovantes »De nouvelles solutions thérapeutiques, de la cible biomacromolécu-laire émergente aux approches originales pour traiter les maladies
Le Thème
« Nouveaux systèmes de vectorisation »Combiner les propriétés d’inclusion de principes actifs, de franchisse-ment de barrières, d’adressage et de relar-gage en milieu vivant
Le Thème
« Outils de bio-détection et bio-quantification »Des dispositifs analytiques originaux pour la détection de cibles, de l’ion au micro-organisme en milieu complexe
Zoom sur... La Chémobiologie
-
Équipes
L'Équipe « COMET »
« COMET » développe la conception rationnelle, la synthèse et/ou l'extraction de composés à forte diversité/complexité comme nouveaux agents thérapeutiques et outils moléculaires pour la pénétration cellulaire ou la détection de biomolécules, actifs in vivo.L'Équipe « NOVA »
« NOVA » utilise des acides nucléiques fonctionnels comme éléments de reconnaissance pour des applications thérapeutiques ou diagnostiques, comme la sélection d'oligonucléotides, ou le développement de dispositifs d'analyses et de nanovecteurs.Les Services
-
Productions
Les Publications
La Vulgarisation
Les JSM
Zoom sur... La 12ème JSM (15 juin 2023)
Le DPM organise des journées scientifiques consacrées au médicament. L'objectif est de rassembler les spécialistes académiques et industriels autour d'une thématique. 2023 : Apports de la Chimie Click et de la Lumière en Chemobiologie
-
Partenariats
Les Formations
Les Consortiums
Les Financements
Zoom sur... L'environnement Grenoblois
Le DPM est un acteur central sur le bassin grenoblois en chimie, biologie et santé, lié au CHU Grenoble Alpes et à de nombreuses autres organisations : Pole de Recherche CBS, ICMG, Labex ARCANE, EUR CBH, Institut Carnot Polynat, Réseau GREEN.
Article
- Projet
- Farid OUKACINE,
- Titre
- Focusing and Mobilization of Bacteria in Capillary Electrophoresis
-
[Full paper
 ]
] - Auteurs
- F. Oukacine, L. Garrelly, B. Romestand, D. M. Goodall, T. Zou, H. Cottet
- Edition
- Anal. Chem. 2011, 83, 1571-1578.
- Année
- 2011
- Résumé
- An isotachophoretic method has been developed for mobilizing and focusing bacteria. This allows quantification and detection of bacteria in a narrow zone. Very good linearity was obtained for Micrococcus lysodeikticus (also called Micrococcus luteus, studied as a model of Gram+ bacteria) in the range of 0.4 x 10(8) cells/mL to 2.9 x 10(8) cells/mL, with correlation coefficients for peak height and peak area as a function of cell concentration of 0.999 and 0.998, respectively. This method is usable on both bare and hydroxypropyl cellulose coated fused silica capillaries. The best results were obtained using 13.6 mM Tris, 150 mM boric acid as terminating electrolyte, and 4.5 mM Tris, 50 mM boric acid, and 3.31 mM HCl as leading electrolyte. With a 33.5 cm x 100 mu m i.d. capillary, short migration times were obtained while maintaining very low electrical current in order to minimize any Joule heating and lysis of the bacteria. A UV area imaging detector (ActiPix D100, Paraytec) was used with a 109 cm x 100 mu m i.d. capillary having three loops and four detection windows to monitor the migration behavior of M luteus and to show the stability of the zone of the focused bacteria along the capillary. Similar results were obtained for Erwinia carotovora (a model of Gram bacteria), and for Enterobacter cloacae and Vibrio splendidus.




 Annuaire
Annuaire Contact
Contact Plan d'accès
Plan d'accès ENG
ENG Login
Login


