- ⌂
-
Le DPM
Les Equipements
L'Environnement
Zoom sur... Le Bâtiment André Rassat
Nommé d'après une figure tutélaire de la chimie grenobloise, ce bâtiment est recouvert d’une double peau en feuille métallique qui apporte une protection thermique sur 3 côtés et crée une unité architecturale favorisant l'intégration parmi les arbres du site.
-
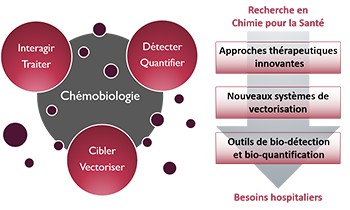
Thématiques
Le Thème
« Approches thérapeutiques innovantes »De nouvelles solutions thérapeutiques, de la cible biomacromolécu-laire émergente aux approches originales pour traiter les maladies
Le Thème
« Nouveaux systèmes de vectorisation »Combiner les propriétés d’inclusion de principes actifs, de franchisse-ment de barrières, d’adressage et de relar-gage en milieu vivant
Le Thème
« Outils de bio-détection et bio-quantification »Des dispositifs analytiques originaux pour la détection de cibles, de l’ion au micro-organisme en milieu complexe
Zoom sur... La Chémobiologie
-
Équipes
L'Équipe « COMET »
« COMET » développe la conception rationnelle, la synthèse et/ou l'extraction de composés à forte diversité/complexité comme nouveaux agents thérapeutiques et outils moléculaires pour la pénétration cellulaire ou la détection de biomolécules, actifs in vivo.L'Équipe « NOVA »
« NOVA » utilise des acides nucléiques fonctionnels comme éléments de reconnaissance pour des applications thérapeutiques ou diagnostiques, comme la sélection d'oligonucléotides, ou le développement de dispositifs d'analyses et de nanovecteurs.Les Services
-
Productions
Les Publications
La Vulgarisation
Les JSM
Zoom sur... La 12ème JSM (15 juin 2023)
Le DPM organise des journées scientifiques consacrées au médicament. L'objectif est de rassembler les spécialistes académiques et industriels autour d'une thématique. 2023 : Apports de la Chimie Click et de la Lumière en Chemobiologie
-
Partenariats
Les Formations
Les Consortiums
Les Financements
Zoom sur... L'environnement Grenoblois
Le DPM est un acteur central sur le bassin grenoblois en chimie, biologie et santé, lié au CHU Grenoble Alpes et à de nombreuses autres organisations : Pole de Recherche CBS, ICMG, Labex ARCANE, EUR CBH, Institut Carnot Polynat, Réseau GREEN.
Article
- Projet
- Martine DEMEUNYNCK, Isabelle BAUSSANNE, Jean-Francois LEFEBVRE,
- Titre
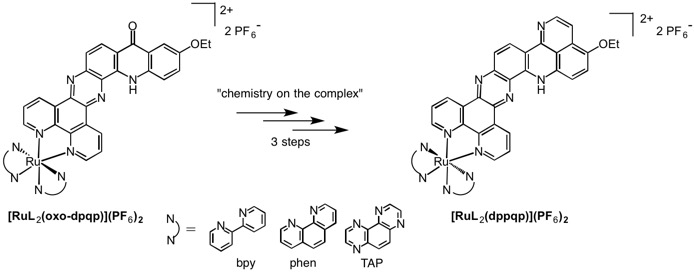
- Synthesis of three series of ruthenium tris-diimine complexes containing acridine-based pi-extended ligands using an efficient “chemistry on the complex” approach
-
[Full paper
 ]
] - Auteurs
- Jean-François Lefebvre, Dounia Saadallah, Philipp Traber, Stephan Kupfer, Stefanie Gräfe, Benjamin Dietzek, Isabelle Baussanne, Julien De Winter, Pascal Gerbaux, Cécile Moucheron, Murielle Chavarot-Kerlidou, Martine Demeunynck
- Edition
- Dalton Trans. 2016, 45, 16298 – 16308
- Année
- 2016
- Résumé
- The preparation and characterization of three series of novel ruthenium (II) complexes are reported, each series differing by the nature of the ancillary ligands (2,2’-bipyridine – bpy, 1,10-phenanthroline – phen or 1,4,5,8-tetraazaphenanthrene – TAP). The third ligand was either the heptacyclic heterocycle dipyrido[3,2-a:2’,3’-c]quinolino[3,2-h]phenazine (dpqp) substituted at position 12 by an hydroxyl (oxo), 2,2-dimethoxyethylamine (DMEA) or halogeno (Cl or Br) substituent, or the octacyclic dipyrido[3,2-a:2’,3’-c]pyrido[2,3,4-de]quinolino[3,2-h]phenazine (dppqp), prepared by a multi-step “chemistry on the complex” strategy from [RuL2(oxo-dpqp)](PF6)2 . The three steps, halogenation, substitution by a dimethoxyethylamino group and cyclization in trifluoroacetic acid, were performed in reasonable to high yields depending on the nature of the ancillary ligands. Isolation and purification processes were facilitated by the ability to switch the solubility of the complex from aqueous to organic solvents, depending on the counter-ion. All the new complexes were fully characterized; in particular their absorption properties were compared by UV-vis spectroscopy. Finally, stacking properties induced by these extended ligands were studied by 1H NMR studies and quantum chemical calculations.




 Annuaire
Annuaire Contact
Contact Plan d'accès
Plan d'accès ENG
ENG Login
Login



