- ⌂
-
Le DPM
Les Equipements
L'Environnement
Zoom sur... Le Bâtiment André Rassat
Nommé d'après une figure tutélaire de la chimie grenobloise, ce bâtiment est recouvert d’une double peau en feuille métallique qui apporte une protection thermique sur 3 côtés et crée une unité architecturale favorisant l'intégration parmi les arbres du site.
-
Thématiques
Le Thème
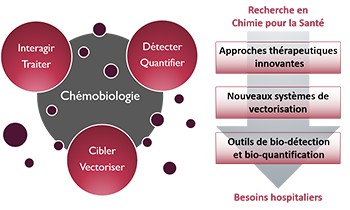
« Approches thérapeutiques innovantes »De nouvelles solutions thérapeutiques, de la cible biomacromolécu-laire émergente aux approches originales pour traiter les maladies
Le Thème
« Nouveaux systèmes de vectorisation »Combiner les propriétés d’inclusion de principes actifs, de franchisse-ment de barrières, d’adressage et de relar-gage en milieu vivant
Le Thème
« Outils de bio-détection et bio-quantification »Des dispositifs analytiques originaux pour la détection de cibles, de l’ion au micro-organisme en milieu complexe
Zoom sur... La Chémobiologie
-
Équipes
L'Équipe « COMET »
« COMET » développe la conception rationnelle, la synthèse et/ou l'extraction de composés à forte diversité/complexité comme nouveaux agents thérapeutiques et outils moléculaires pour la pénétration cellulaire ou la détection de biomolécules, actifs in vivo.L'Équipe « NOVA »
« NOVA » utilise des acides nucléiques fonctionnels comme éléments de reconnaissance pour des applications thérapeutiques ou diagnostiques, comme la sélection d'oligonucléotides, ou le développement de dispositifs d'analyses et de nanovecteurs.Les Services
-
Productions
Les Publications
La Vulgarisation
Les JSM
Zoom sur... La 12ème JSM (15 juin 2023)
Le DPM organise des journées scientifiques consacrées au médicament. L'objectif est de rassembler les spécialistes académiques et industriels autour d'une thématique. 2023 : Apports de la Chimie Click et de la Lumière en Chemobiologie
-
Partenariats
Les Formations
Les Consortiums
Les Financements
Zoom sur... L'environnement Grenoblois
Le DPM est un acteur central sur le bassin grenoblois en chimie, biologie et santé, lié au CHU Grenoble Alpes et à de nombreuses autres organisations : Pole de Recherche CBS, ICMG, Labex ARCANE, EUR CBH, Institut Carnot Polynat, Réseau GREEN.
Article
- Projet
- Martine DEMEUNYNCK, Isabelle BAUSSANNE,
- Titre
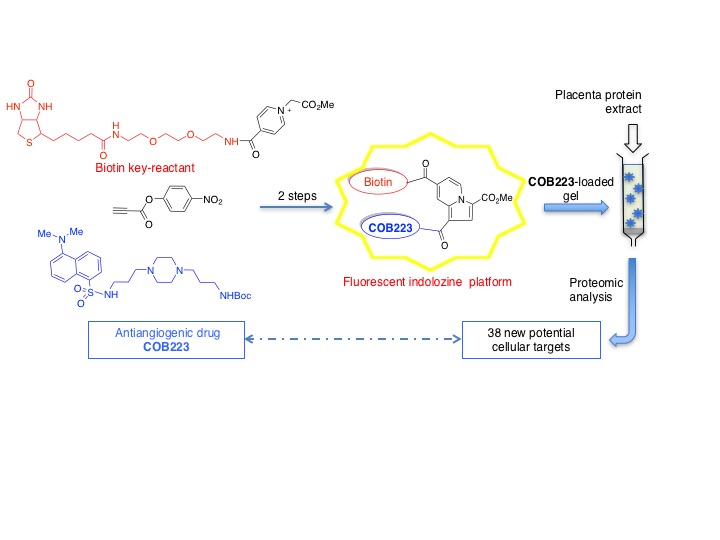
- Indolizine-based scaffolds as efficient and versatile scaffolds for drug target identification – Synthesis and study of biotin-tagged anti-angiogenic drugs
- Auteurs
- Marie Arvin-Berod, Simon Bonte, Agnès Desroches-Castan, Sabine Brugière, Yohann Couté, Laurent Guyon, Jean-Jacques Feige, Isabelle Baussanne and Martine Demeunynck
- Edition
- ACS Omega, 2017, 2, 9221-9230
- Année
- 2017
- Résumé
- We describe the synthesis of polyfunctional scaffolds based on a fluorescent indolizine core derivatized with various orthogonal groups. The scaffold was used to prepare drug-biotin conjugates that were then immobilized onto avidin-agarose for affinity chromatography. More specifically, the anti-angiogenic drug COB223, whose mechanism of action remained unclear, was chosen as a proof-of-concept drug. Separation of COB223-interacting proteins from human placental extracts unveiled unanticipated protein targets belonging to the family of regulatory RNA-binding proteins, which opens the way to new hypotheses on the mode of action of this anti-angiogenic drugs




 Annuaire
Annuaire Contact
Contact Plan d'accès
Plan d'accès ENG
ENG Login
Login



